Gemcitabine Drug: Indication, Dosage, Precaution, Side Effect , Storage, Category Type and corresponding Brands - www.genericdrugscan.com
Gemcitabine
Drug Status in USA : ApprovedDrug Status in Canada : Approved
Indication : Why is this medication prescribed?
Adenocarcinoma of the pancreas - non resectable stage II, stage III and stage IV. Advanced non small cell lung cancer. Bladder cancer. Breast cancer and ovarian carcinoma.Contraindication: What are the special precautions to be followed?
Under following conditions, use of this medication is inadvisable. Its use should be withhold as it would cause harm to the patientHypersensitivity. Concurrent radical radiotherapy, pregnancy and lactation.
Other Precaution to be observed while taking this medication
Patients should be monitored with complete blood count before each dose. Renal and hepatic functions should be evaluated before initiation of therapy and periodically thereafter. May impair ability to drive or operate machinery.What are possible side effects of this medication ?
Myelo-suppression (leukopenia, anaemia, thrombocytopenia), parasthesias, nausea, anorexia, headache, fever, rash, renal/hepatic impairment, vomiting, dyspnoea, peripheral, oedema, flu like syndrome, asthenia, cough, chills, interstitial pneumonia, pulmonary oedema. Proteinuria, haematuria and haemolytic uraemic syndrome. OesophagitisDrug Category/Class
- Antimetabolites, Antineoplastic
- Enzyme Inhibitors
- Immunosuppressive Agents
- Radiation-Sensitizing Agents
- Pyrimidine Analogues
- Antimetabolites
- Antineoplastic Agents
- Antineoplastic and Immunomodulating Agents
- Antiviral Agents
- Pyrimidine analogues
| Prescribed | Gemcitabine is indicated for the treatment of advanced ovarian cancer that has relapsed at least 6 months after completion of platinum-based therap... |
| Weight : | 263.1981 |
| Structure | Gemcitabine |
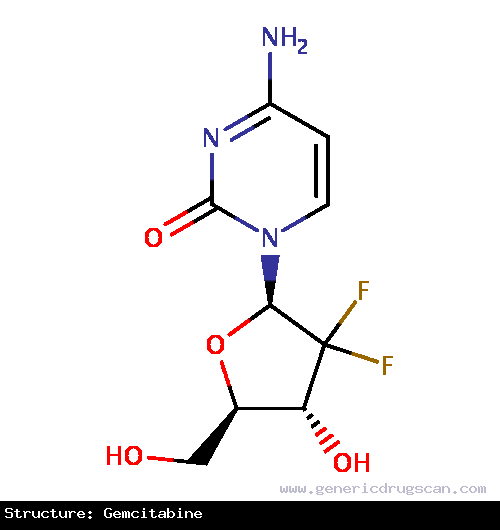 | |
| Formula | C9H11F2N3O4 |